Coronavirus
Singapore may get non-mRNA Novavax vaccine by year-end
Vaccine still in phase 3 trials, but late-stage data from US shows more than 90% efficacy
Sign up now: Get ST's newsletters delivered to your inbox
Singapore has signed advance purchase agreements with American biotechnology company Novavax to secure its protein-based Covid-19 vaccine, with supplies possibly arriving before the end of the year.
The Ministry of Health (MOH) signed the agreements with Novavax in January, Health Minister Ong Ye Kung said yesterday at a virtual media conference held by the multi-ministry task force tackling Covid-19.
He added that MOH has been looking for vaccines of good quality, which are safe and effective, to be part of the national vaccination programme, and noted that Novavax has recently showed encouraging results.
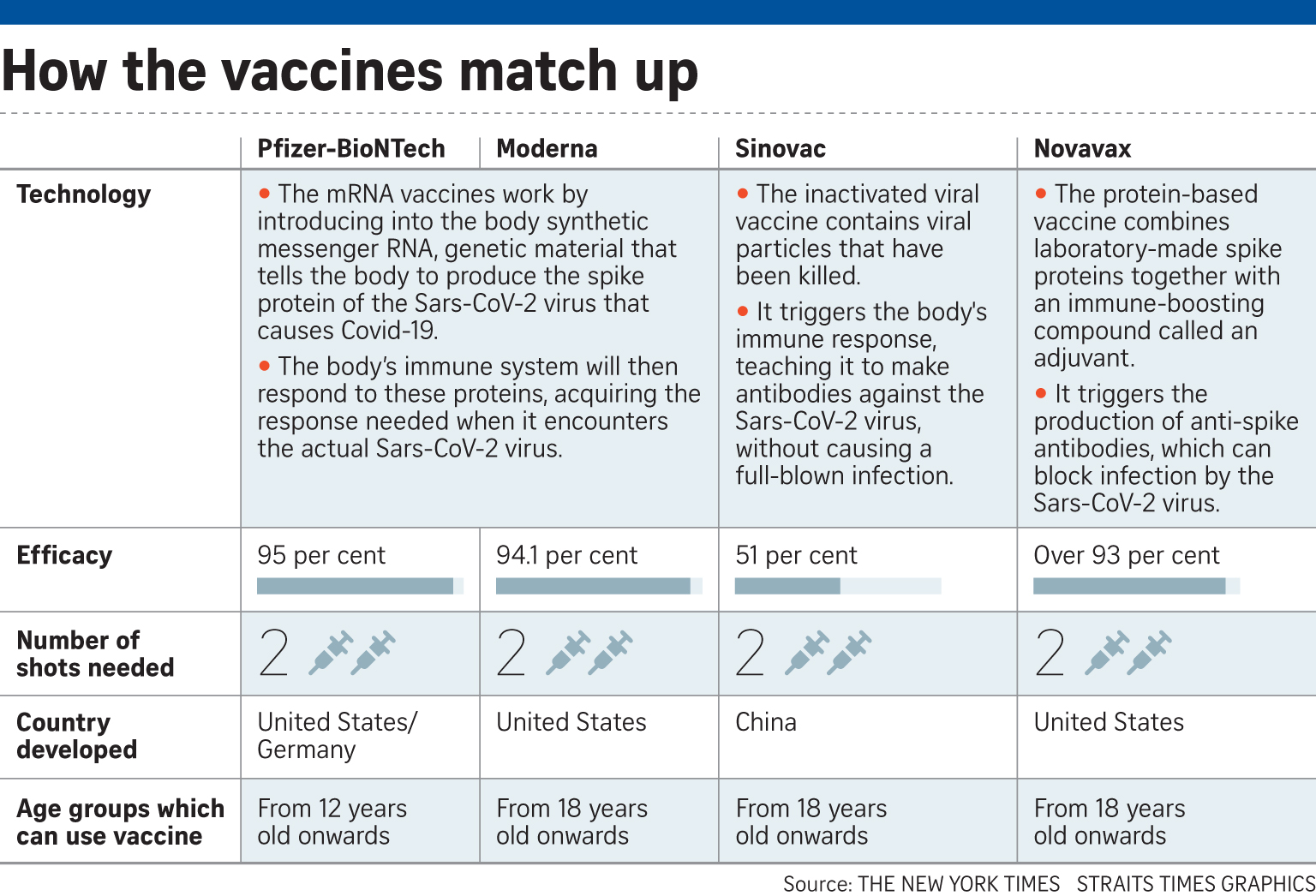
Novavax has been shown to be more than 90 per cent effective against a variety of Covid-19 variants, based on late-stage data from its clinical trial in the United States.
Mr Ong said shipments of Novavax's vaccines will not be ready so soon as it is still undergoing phase three clinical trials.
"In the meantime, please consider mRNA vaccines, they work very well," he added.
Currently, only the Pfizer-BioNTech and Moderna vaccines, which use mRNA technology, are offered under Singapore's vaccination programme. These vaccines teach cells to make a protein that prompts an immune response.
Novavax, a protein-based vaccine, uses a laboratory-made version of the Sars-CoV-2 spike protein to stimulate an immune response.
Like the Pfizer and Moderna vaccines, it also requires two shots, spaced apart.
Earlier this month, a study of nearly 30,000 volunteers in the US and Mexico put Novavax on track to file for emergency authorisation in the US and elsewhere in the third quarter of the year, the company said on June 14.
Novavax added that its vaccine candidate was more than 93 per cent effective against the predominant variants of Covid-19 that have been of concern among scientists and public health officials.
Protein-based vaccines are a conventional approach that uses purified pieces of the virus to spur an immune response. Long-established vaccines against whooping cough and shingles employ this approach.
In Singapore, some people have opted for the Sinovac Covid-19 vaccine, which uses deactivated coronaviruses to trigger an immune response - another conventional vaccine technology.
Sinovac was recently allowed for use here under the Special Access Route framework to enhance the country's overall vaccination coverage, though it is not authorised for use under the Pandemic Special Access Route and is not included in the national vaccination programme.
Some of the 24 private healthcare clinics that provide the Sinovac vaccine have seen long queues, with some people choosing to pay for a vaccine that uses a more conventional technology, even though studies have found that it is less effective than the mRNA technology.
Yesterday, Singapore's director of medical services Kenneth Mak was also asked about ancedotal accounts of some people who chose the Pfizer-BioNTech vaccine over the Moderna jab, due to a supposedly higher rate of adverse reactions associated with the Moderna jab.
Noting that this observation has not been borne out by data, he said: "We have been looking at adverse events for all vaccines... and at this point in time, there is no signal to suggest a higher adverse event rate associated with Moderna."
He added: "We have been therefore encouraging all to get whichever vaccine is available, given that supplies come in at different points in time... Neither is better than the other."